Sage Therapeutics Inc.’s drug helped improve depressive symptoms after three days in a late-stage study, but investor concerns over how long the treatment’s effect would last sent the drug developer’s shares down 17% on February 16.
Roivant Sciences launched the new subsidiary Hemavant, which hit the ground running with a licensing agreement from Eisai for RVT-2001, a potential first-in-class small molecule SF3B1 modulator.
Prior COVID offers less protection vs Omicron; mRNA booster shot efficacy declines within months
BNT162b2 (Pfizer and BioNTech), CDC, Clinical Data, Coronavirus Disease 2019 (COVID-19) Reinfections, COVID-19 booster shots, Covid-19 Data, COVID-19 shots, COVID-19 Vaccines, Hospitalized COVID-19 Patients, Immune Response, Messenger RNA (mRNA) Vaccines, mRNA-1273/Moderna COVID-19 Vaccine (Moderna), New England Journal of Medicine, Omicron (B.1.1.529) (South Africa), Omicron BA.2, R&DThe immune response to COVID-19 helps protect against reinfection, but that protection is weaker against Omicron than it was against earlier variants of the coronavirus, according to new data. In other news, protection provided by booster shots of the mRNA vaccines from Moderna Inc. or Pfizer Inc. and partner BioNTech SE starts waning quickly, according to data published in Feb. 11th’s Morbidity and Mortality Weekly Report from the U.S. Centers for Disease Control and Prevention (CDC).
Pardes Biosciences Inc. said on February 14 the company expects to start a mid-to-late-stage trial of its experimental COVID-19 antiviral pill by mid-2022, following promising data from early human testing.
Pfizer Postpones EUA in Kids Under 5, Will Wait for Full Data
BNT162b2 (Pfizer and BioNTech), Children, Clinical Data, COVID-19 booster shots, Covid-19 Data, COVID-19 shots, COVID-19 Vaccines, Data Monitoring Committee (DMC), Delta Variant (B.1.617.2; India), Emergency Use Authorization (EUA), FDA, Mayo Clinic, NBC, Omicron (B.1.1.529) (South Africa), Survey Data, Vaccines and Related Biological Products Advisory CommitteeOn the day the public and members of the U.S. Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee were going to get a look at Pfizer and BioNTech’s COVID-19 vaccine data for children under 5 years old, the companies pulled the Emergency Use Authorization (EUA) submission.
The latest results from Genentech’s proposed eye disease treatments are demonstrating strong potential to help reduce treatment frequencies and deliver improved outcomes.
Gilead says COVID drug remdesivir shows antiviral activity against Omicron, other variants
Antivirals, Clinical Data, Clinical Trials, Covid-19 Data, COVID-19 Therapeutics, Covid-19 Variants, Delta Variant (B.1.617.2; India), Gilead, Omicron (B.1.1.529) (South Africa), Omicron BA.2, R&D, Remdesivir, TherapeuticsGilead Sciences Inc.’s antiviral drug remdesivir showed antiviral activity against Omicron, Delta and other variants of the coronavirus in laboratory studies, the company said on February 11.
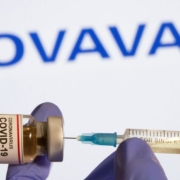
Novavax Inc. said on February 10 the company’s two-dose vaccine was 80% effective against COVID-19 in a late-stage trial testing the shot in teens aged 12 to 17 years.
GSK-Vir therapy has neutralizing activity against Omicron sub-variant, data shows
Antibodies, Broadly Neutralizing Antibodies (bNAbs), Clinical Data, Clinical Trials, Columbia University, Covid-19 Data, COVID-19 Studies, GlaxoSmithKline, Monoclonal Antibodies, Omicron BA.2, R&D, Researchers, Sotrovimab, Vir BiotechnologyAn antibody-based COVID-19 therapy developed by GSK and Vir Biotechnology retains neutralizing activity against the emerging BA.2 form of the Omicron coronavirus variant, according to data from a laboratory study cited by Vir on February 10.
COVID Update: Revisiting the Lab-Leak Theory and Under 5 Vaccination
Beijing, CDC, Children, China, Clinical Data, Clinical Trials, Covid-19 Data, COVID-19 Infections, COVID-19 Origins, COVID-19 Vaccinations, Emergency Use Authorization (EUA), GlaxoSmithKline, Heart Disease, Heart Problems, Janssen, Johnson & Johnson, Neutralizing Antibodies, New England Journal of Medicine, Omicron (B.1.1.529) (South Africa), Omicron BA.2, R&D, Scripps Research Institute, Vir BiotechnologyJane Qiu, a freelance science writer based in Beijing, published an overview of her efforts to investigate the lab-leak theory for the origin of COVID-19. In another update, the FDA’s Vaccines and Related Biological Products Advisory Committee is scheduled to meet on February 15 to discuss an amended Emergency Use Authorization for the Pfizer-BioNTech COVID-19 vaccine for children 6 months through 4 years of age.







 Reuters
Reuters