Through a glass, (more) clearly
Through a glass, (more) clearly
The greatest power of the new marketing technologies available to pharma brands may be their capacity to bring the beliefs and questions of real patients and HCPs into sharper view.
By Joshua Slatko • [email protected]
No matter how complicated our newest marketing technologies get under the hood, the problems they aim to solve and the questions they attempt to answer remain simple. And the simplest question of all, going back to the very beginning of marketing, is: how do my customers, and potential customers, view my product? Armed with all the growing power of big data, artificial intelligence, vast processing power, machine learning, and all the rest, brand teams are finding that they can answer that first simple question to a degree of granularity and detail unimaginable to previous generations, or even to themselves just a few years ago. Want to know what patients are thinking or saying about your brand? Want to know how much HCPs know about it? Want to know what questions either are asking? The answers, thanks to the tools profiled below and others like them, are just a click away.
PatientMetRx

PatientMetRx’s Patient Opinion Maps help users track patterns and trends in patient views about a medicine.
Talking Medicines’ PatientMetRx AI platform is designed to sift through millions of patient conversations online to determine how patients are experiencing their medicines in the real world. The platform is driven by AI models that use machine learning and natural language processing to provide science-based insights that cut through the noise from social media. PatientMetRx uses real-time continuous sourcing from public social platforms and private platforms with permissions.
“We are agnostic as to where those insights come from so long as we can find genuine patient experience within it,” says Jo Halliday, CEO and co-founder of Talking Medicines. “For example, Talking Medicines are able to source unstructured text from the large social media firehoses through to message boards and specific special interest groups. We can also run a client’s unstructured data source through our models.”
According to company leaders, the platform’s proprietary Patient Data Classifier and Named Entity Recognition models pull out patient voices from hundreds of millions of social posts at an accuracy of 95 percent. This allows for real-time tracking of Patient Confidence, Patient Opinions, Patient Personas, and Patient Language on the PatientMetRx online dashboard, allowing users to quickly and effectively access their patient insights at a medicine brand level. “At the touch of a button, users can access real-time, structured, and unlimited data that has been distilled into clear insights designed to help healthcare agencies win and retain pharma business through excellence in their patient strategies,” Halliday says.
The platform can be accessed on a subscription basis through buying medicine brand seats. “We encourage usage and do not charge by user seat within an agency realizing that multiple stakeholders need to be able to access the lived experience of patients when, for example, working on a pitch,” Halliday says. PatientMetRx differs from other social listening tools, its creators say, in that the platform does not require the client to write queries to see results. “The AI has already filtered potentially millions of conversations to find the Patient Voice. The tech homes in on the direct patient experience about medicines, and the context around this. As with panning for gold, we only serve the relevant insights rather than full query results.”
Insights can be accessed in a variety of ways through the features within the platform. For example, there is a Patient Confidence Score that calculates the brand health for a medicine and provides a summation of patient experience. Further analysis of insights can be made through the Patient Feed and Patient Opinion Map features, which offer structured ways to follow patterns and trends and search specific topics that patients have talked about.
How did all this come to be? “Talking Medicines was established as a mission-led business to help pharmaceutical marketing industry put patients right in the center of everything they do,” Halliday says. “To achieve this, we had to find a way to connect the patient voice to pharma while considering the highly regulated nature of the industry. Three major changes in the medicines market persuaded us to accelerate our capabilities in AI in capturing the patient voice.”
Pre-COVID, Halliday explains, pharma relied heavily on deriving patient insights from healthcare professionals. However, with less reps on the road able to meet face to face with healthcare professionals, there was an evident forced change in the feedback loop. At the same time, as a result of the pandemic, fewer patients were visiting their GPs and more were increasingly turning to social media channels to find peer-to peer-support, alongside voicing their experiences about treatment, conditions, and medicines. Finally, the Talking Medicines team noticed that healthcare marketers were increasingly looking to tech to help with the automation of data analysis as well as the adoption being seen by pharma toward digital transformation and data-driven solutions.
“Having built great credibility and experience in working with pharma companies and healthcare agencies, we realized that there was a market opportunity to use technology to help pharma connect with the true patient voice,” Halliday says. “Pharma outsources patient marketing to healthcare agencies, so partnering with those agencies has been a natural step. We have only ever sold to pharma and healthcare, giving us a deep understanding of needs, opportunities and challenges, as well as a strong domain knowledge around patients, mining data and servicing healthcare clients is strong.”
As the pandemic brought much business as usual to a halt, the Talking Medicines team raised funding and dedicated R&D time to developing the AI models and the early platform. “From the very beginning, compliance and quality have always been high on the agenda for us – a key priority that was driven by the vision for the company in supporting patients by giving them a voice,” Halliday says.
So how might access to PatientMetRx impact a brand team’s awareness of market conditions and subsequent decision making? Talking Medicines tried out a test case last year to find out. In September 2021, an FDA-approved autoimmune medicine was at the center of a safety concern, which culminated in a black box warning. “We wanted to use PatientMetRx to see how this regulatory event would impact patient confidence and opinion,” Halliday told Med Ad News. “Running the data through our platform, we looked at what terms could be associated with this event, how many opinion posts were talking about the black box warning, and which terms and in what quantity these terms were mentioned in opinion posts. Having identified the key themes (by using tags) we filtered our opinion map by these themes, allowing a more focused overview of patient sentiment. We found that while the FDA and safety concerns did arise in our analysis, they were not common. In fact, contrary to popular belief, we found that positive appraisals of the medicine were more common and people spoke in positive terms.” The team was quickly able to establish the impact of the FDA warning on patient perception both contextually and from a sentiment analysis perspective, insights that could be used to inform specific communication strategy and language in the face of a regulatory issue. “And, just as important, our ability to analyze this data was 80 percent faster than a conventional Excel-based analysis.”
Looking forward, the Talking Medicines leadership plans to enhance the depth and breadth of PatientMetRx both within its current market of healthcare advertising agencies as well as the adjacent industries of insurance and pharmacy. “The future involves prediction, as well as more depth of socio-economic filters,” Halliday says. “We have heard the need for health care to improve the way they understand and market to subgroups of patients whether by age, gender, location, [or] ethnicity.”
Most importantly, PatientMetRx will be staying focused entirely on the patient. “Our view of only looking at patients is what differentiates PatientMetRx from other data analytics tools,” Halliday says. “This laser focus gives us a level of accuracy and sophistication that would be spread thinly if we applied it more broadly. It is one part of the jigsaw, as other tools service needs from the healthcare professional/HCP and other voices, but we have made it our purpose to provide gold standard insights on the patient voice.”
Scientific Awareness
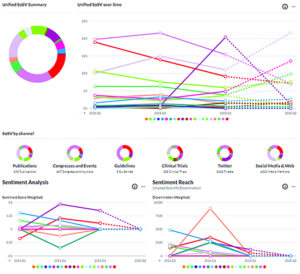
Veeva’s Link for Scientific Awareness app helps measure awareness and sentiment among HCPs by calculating share of scientific voice across a broad range of sources.
Veeva’s Link for Scientific Awareness app, a part of its broader Link platform, measures awareness and sentiment among HCPs regarding specific medicines. The app’s real-time dashboards track the share of scientific voice across all relevant scientific and digital sources, including publications, congresses, clinical guidelines, social media, and professional associations. On the back end, more than 100 human data curators with deep scientific and clinical knowledge are on hand to measure sentiment, which helps users understand how the scientific and clinical community “feels” about different treatments.
“Understanding the actions and sentiments of scientific experts and prescribers has become an essential part of the industry’s efforts to strengthen field engagement and drive better patient outcomes,” says Kilian Weiss, general manager of Veeva Link. “This expanded need for real-time intelligence applications drove us to launch Link for Scientific Awareness as the first in our four new Link data applications that include Key Accounts, Medical Insights, and Link+ for agile workflow and relationship management.”
The push for the Scientific Awareness app, Weiss told Med Ad News, came not from the brand marketing side but from medical affairs teams. Unlike their commercial counterparts who rely on market share and sales data to determine performance and impact, medical affairs cannot tie its performance to commercial measures. So a bit more creativity in measurement is necessary.
“After hosting multiple brainstorming sessions with different medical teams, we defined three metrics that are valuable to track,” Weiss explains. “Scientific share of voice is an important measure of visibility and awareness, while sentiment and sentiment reach allows those teams to capture the human factor. For example, how the medical and scientific community interprets their scientific platform and what’s the prevalence of that opinion. It’s not just Medical Affairs that find our product useful, though; commercial teams have also been excited to see how their campaigns are reflected through a new, unbiased external lens.”
That external lens is able to dig through an enormous amount of relevant scientific data almost instantaneously. ASCO, ESMO, and other congresses now consistently publish agendas, speaker rosters, and scientific presentations online. The app can also pull data from ClinicalTrials.gov, PubMed, Twitter, YouTube, and elsewhere. “We can track and measure this data as soon as it’s published,” Weiss explains. “Our dashboards help medical teams make sense of all this data by visualizing where their product stands in comparison to competitors, as well as showing what data channels and people are the largest contributors to their scientific share of voice. It’s valuable to track the progression of your scientific share of voice over time, and you can group the data into categories such as publications, clinical trials, social media, and so forth.”
Veeva’s Scientific Awareness app is growing up alongside another online phenomenon, the HCP digital opinion leader. “Interestingly, digital opinion leaders may not be the true experts in the field, yet they drive visibility and engagement by constantly being digitally active and prolific,” Weiss says. “Now that there’s a clear trend of the increasing importance of digital opinion leaders, our customers are exploring how to best engage this customer group. Even though this trend was accelerated by the pandemic, it’s clear that it’s likely to stay.”
So what sorts of questions are users asking with the help of the app? “What congresses are going to be impactful?” Weiss says. “What publications should be considered? Where is my audience and where should the focus be?” And the answers have already been used to brand benefit. In one case, a top 10 pharmaceutical company was able to increase by 50 percent its share of scientific voice and keep it for one of their products in just two quarters, which was especially significant as this was during its brand’s launch. When analyzing the competitive landscape and underlying sentiment data for this class of therapy, the team found that other companies were focusing on driving diagnostic tool awareness ahead of focusing on the therapy they were launching. They also uncovered clear HCP concerns about outpatient center preparedness like logistical barriers to carrying radiolabeled therapy that needed to be addressed early to ensure strong therapy adoption. With this knowledge in hand, they were able to fine-tune communications accordingly and gain that impressive bounce in voice.
Just as with PatientMetRx, the future of the Scientific Awareness app lies, at least in part, in a predictive capability. “Our goal is to make it even easier for our customers to use Link applications in their decision-making process,” Weiss says. “The next step is to use all this data across different channels and products and propose intelligent recommendations on what should be done to achieve the outcome the user is seeking.”
Veeva is also exploring the possibility of applying advanced technologies and powerful analytical engines to structured and unstructured data, a trend already well under way throughout the healthcare industry. “Our customers are faced with a real challenge of trying to integrate insights across the multitude of data sources available, both their own and external,” Weiss told Med Ad News. “Artificial intelligence, machine learning, and natural language processing can help significantly accelerate insight integration and create ‘meta’ insights, which previously hasn’t been possible. We are still in the nascent stage of leveraging data to show how products and therapies are making an impact. Share of scientific voice and sentiment are precursors to true impact. We want to be able to demonstrate that customers’ activities are making a difference in increasing access to treatments for patients that need them to improve and extend patient lives.”
 |
Josh Slatko is contributing editor of Med Ad News and PharmaLive.com. |










