U.S. FDA approves Eisai, Biogen’s Alzheimer’s drug
U.S. FDA approves Eisai, Biogen’s Alzheimer’s drug
Jan 6 (Reuters) – The U.S. Food and Drug Administration approved Eisai Co Ltd’s (4523.T) and Biogen Inc’s (BIIB.O) Alzheimer’s drug lecanemab for patients in the earliest stages of the mind-wasting disease, the agency said on Friday.
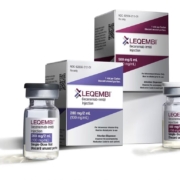
The drug, to be sold under the brand Leqembi, belongs to a class of treatments that aims to slow the advance of the neurodegenerative disease by removing sticky clumps of the toxic brain protein known as beta amyloid.
Nearly all previous experimental drugs using the same approach had failed.
Initial access to the drug likely will be limited by a number of factors including reimbursement decisions from Medicare, the U.S. government insurance program for Americans aged 65 and older who represent some 90% of individuals likely to be eligible for Leqembi.
Our Standards: The Thomson Reuters Trust Principles.
Source: Reuters