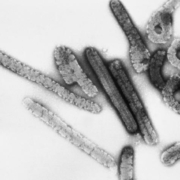
The FDA approved bluebird bio’s lentiviral vector (LVV) eli-cel Friday as the first therapy to slow the progression of neurologic dysfunction in juvenile boys with early, active cerebral adrenoleukodystrophy (CALD).
Featured Posts
 FDA accelerated approval: implications for treatment innovation in oncology - Recent U.S. policy changes and the introduction of the Inflation Reduction Act (IRA) have injected a sense of urgency within pharma companies for greater cross functional cooperation. The traditional route that drugs take through the development process from bench to patient needs a radical rethink. Any pressure on how drugs are assessed through the AA route will further ramp up the urgency within pharma to align internally.
FDA accelerated approval: implications for treatment innovation in oncology - Recent U.S. policy changes and the introduction of the Inflation Reduction Act (IRA) have injected a sense of urgency within pharma companies for greater cross functional cooperation. The traditional route that drugs take through the development process from bench to patient needs a radical rethink. Any pressure on how drugs are assessed through the AA route will further ramp up the urgency within pharma to align internally.Ghana has declared the end of an outbreak of Marburg virus, a highly infectious disease similar to Ebola, that was confirmed in July, the president’s office said on Monday.
Teva Pharmaceutical Industries expects to finalize an opioid settlement in the United States by year-end and start paying in 2023, its chief executive said on Sunday, while confirming he was unlikely to renew his contract next year.
A senior Chinese health official advised people to avoid contact with foreigners to prevent monkeypox infection after the first known case of the virus on mainland China was reported.
U.S. President Joe Biden said in an interview aired on Sunday that “the pandemic is over,” even though the country continues to grapple with coronavirus infections that kill hundreds of Americans daily.
The FDA approved Mallinckrodt Pharmaceuticals’ Terlivaz (terlipressin) in adults with hepatorenal syndrome (HRS), making it the only FDA-approved drug for that indication in the U.S.
Five patients with systemic lupus erythematosus (SLE) are in remission after receiving CAR T cell therapy, according to a study published Thursday in Nature Medicine.
France’s national health body warned on Friday of a resurgence of COVID-19 cases in the country, and urged people to continue to get vaccinated to protect themselves against the virus.
Two COVID-19 antibody therapies are no longer recommended by the World Health Organization (WHO), on the basis that Omicron and the variant’s latest offshoots have likely rendered them obsolete.
Europe’s medicines regulator has backed using AstraZeneca’s preventative COVID-19 therapy as a treatment for the disease and also endorsed another medicine as preventative option for another common virus.